NOBLE Study Brings Advanced Prostate Cancer Imaging to Rural Australia
First Patient Dosed. NOBody LEft behind.
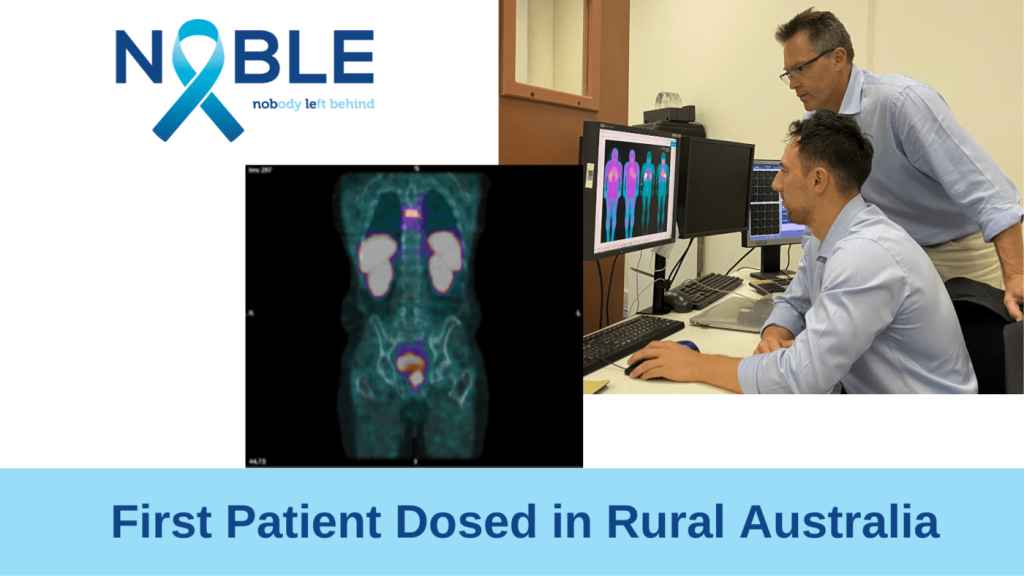
The first Australian patient has been dosed within the NOBLE Registry, an international study that aims to improve access for men to state-of-the-art prostate cancer imaging tools. The NOBLE Registry is co-supported by Telix Pharmaceuticals and the Oncidium foundation.
The NOBLE (Nobody Left Behind) Registry is collecting clinical data to inform the development of 99mTc-iPSMA, an investigational prostate cancer imaging agent that targets PSMA (prostate specific membrane antigen) using single photon emission computed tomography (SPECT). The Australian arm of the NOBLE study is being led by PhD researcher Peter Tually at TeleMed in collaboration with Clinical Professor Nat Lenzo, and Professor Geoff Currie at Charles Sturt University (CSU), and will recruit prostate cancer patients in the Kalgoorlie, Albany, and Geraldton regions in rural Western Australia.
While PSMA-PET imaging is emerging as the new standard of care for prostate cancer diagnosis and staging, having been recently included in the updated National Comprehensive Cancer Network Guidelines®, access is typically limited outside of major cities and in emerging health care systems. A potential alternative is currently being evaluated within the NOBLE Registry using SPECT, which is a predominant imaging modality, particularly for certain countries as well as remote and rural locations.
Nuclear medicine practitioner, Peter Tually, Principal Investigator for the NOBLE Registry study in Australia and Director of TeleMed, a provider of tele-nuclear medicine and paramedic services to rural and remote communities of Western Australia, said, “Our mission is to bring a metropolitan standard of care to the bush and level the playing field where we can. Our involvement in NOBLE takes us a step closer to achieving this, by giving patients local access to the latest prostate cancer imaging tools which could have a profound impact on clinical decisions regarding their treatment.”
The project, according to Professor Currie, “is consistent with CSU leadership in rural health and commitment to addressing social asymmetry, inequity and injustice of health-related outcomes.”
Dr. Christian Behrenbruch, Telix CEO added, “The NOBLE study is an important piece of clinical research which could expedite access to PSMA imaging for patients who might otherwise have to travel long distances or endure significant wait times to access this technology. With the support of investigators such as Peter Tually and Professor Geoff Currie, and their global counterparts, we are recruiting patients at a steady pace, which will pave the way for greater access to PSMA imaging for patients in developing nations, rural and remote areas – first on study use basis, but ultimately as a regulatory approved imaging agent.”
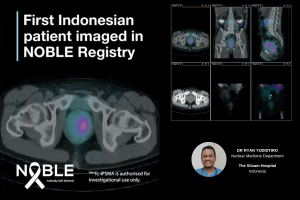
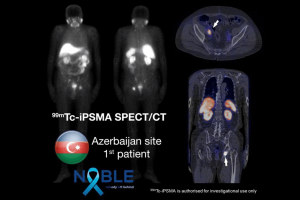
The NOBLE Registry is being conducted at seven sites globally, with a site in Jakarta, Indonesia now added to the study. In addition to Australia and Indonesia, clinical sites are located in Egypt, Mexico, Nigeria, South Africa, and the United Arab Emirates.